Seznamy Atom Structure Of Chlorine
Seznamy Atom Structure Of Chlorine. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. It is very easy to draw cl2 lewis structure. The stability of an element's outer electrons determines its chemical and … Chlorine is a diatomic molecule and contains only two chlorine atoms.
Prezentováno Chlorine Periodic Table And Atomic Properties
05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. How many electrons are in each energy level of the bohr model for chlorine? 22.05.2021 · what is the atomic structure for chlorine? How to draw the atomic model for chlorine.How to draw the atomic model for chlorine.
Thus, it has seventeen protons and seventeen electrons. The atomic number of chlorine is 17; 17), the most common isotope of this element. Chlorine is a diatomic molecule and contains only two chlorine atoms. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. That means there are 17 electrons in a. Thus, it has seventeen protons and seventeen electrons.

Its electron configuration is … 22.05.2021 · what is the atomic structure for chlorine? It is a halogen (17 th group) in the 3 rd period of the periodic table. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. The atomic number of chlorine is 17;. Its electron configuration is …

That means there are 17 electrons in a. It is a halogen (17 th group) in the 3 rd period of the periodic table. The nucleus consists of 17 protons (red) and 18 neutrons (blue). The atomic number of chlorine is 17; Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds.. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl.

05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. It is very easy to draw cl2 lewis structure. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. Chlorine is a diatomic molecule and contains only two chlorine atoms. This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. How many electrons are in each energy level of the bohr model for chlorine? The stability of an element's outer electrons determines its chemical and … 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. How to draw the atomic model for chlorine. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds.

22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. 17), the most common isotope of this element. How many electrons are in each energy level of the bohr model for chlorine? 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. It is very easy to draw cl2 lewis structure. The stability of an element's outer electrons determines its chemical and … Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. That means there are 17 electrons in a. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization.. 22.05.2021 · what is the atomic structure for chlorine?

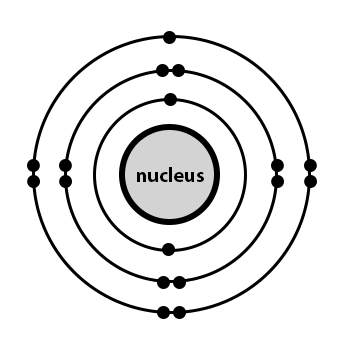
It is very easy to draw cl2 lewis structure. It is a halogen (17 th group) in the 3 rd period of the periodic table. This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). How to draw the atomic model for chlorine. Chlorine is a diatomic molecule and contains only two chlorine atoms. That means there are 17 electrons in a. The atomic number of chlorine is 17; 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization... This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites.

Thus, it has seventeen protons and seventeen electrons. That means there are 17 electrons in a. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 17), the most common isotope of this element. It is very easy to draw cl2 lewis structure. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. The atomic number of chlorine is 17; This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. The stability of an element's outer electrons determines its chemical and … How to draw the atomic model for chlorine. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. Chlorine is a diatomic molecule and contains only two chlorine atoms.
Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure.. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. The stability of an element's outer electrons determines its chemical and … The nucleus consists of 17 protons (red) and 18 neutrons (blue). It is a halogen (17 th group) in the 3 rd period of the periodic table. Thus, it has seventeen protons and seventeen electrons... 22.05.2021 · what is the atomic structure for chlorine?

22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. It is a halogen (17 th group) in the 3 rd period of the periodic table. Chlorine is a diatomic molecule and contains only two chlorine atoms. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl... It is very easy to draw cl2 lewis structure.

Thus, it has seventeen protons and seventeen electrons... The stability of an element's outer electrons determines its chemical and … The atomic number of chlorine is 17; 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

How many electrons are in each energy level of the bohr model for chlorine? The atomic number of chlorine is 17;.. Thus, it has seventeen protons and seventeen electrons.

Chlorine is a diatomic molecule and contains only two chlorine atoms. .. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific.

The nucleus consists of 17 protons (red) and 18 neutrons (blue).. 22.05.2021 · what is the atomic structure for chlorine? How to draw the atomic model for chlorine. Chlorine is a diatomic molecule and contains only two chlorine atoms. That means there are 17 electrons in a. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. The atomic number of chlorine is 17; The nucleus consists of 17 protons (red) and 18 neutrons (blue).. It is very easy to draw cl2 lewis structure.

Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. Chlorine is a diatomic molecule and contains only two chlorine atoms. 22.05.2021 · what is the atomic structure for chlorine? The nucleus consists of 17 protons (red) and 18 neutrons (blue). 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific... Thus, it has seventeen protons and seventeen electrons.

Thus, it has seventeen protons and seventeen electrons... Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. Its electron configuration is … That means there are 17 electrons in a. It is very easy to draw cl2 lewis structure. Thus, it has seventeen protons and seventeen electrons. It is a halogen (17 th group) in the 3 rd period of the periodic table. The stability of an element's outer electrons determines its chemical and … Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure.

Its electron configuration is … 17), the most common isotope of this element. It is a halogen (17 th group) in the 3 rd period of the periodic table. Its electron configuration is … How to draw the atomic model for chlorine. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. The nucleus consists of 17 protons (red) and 18 neutrons (blue). The atomic number of chlorine is 17; 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific.. How to draw the atomic model for chlorine.

Chlorine is a diatomic molecule and contains only two chlorine atoms. The atomic number of chlorine is 17; 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). How many electrons are in each energy level of the bohr model for chlorine? This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. Thus, it has seventeen protons and seventeen electrons.

The atomic number of chlorine is 17; The atomic number of chlorine is 17; Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. It is a halogen (17 th group) in the 3 rd period of the periodic table. Thus, it has seventeen protons and seventeen electrons. The stability of an element's outer electrons determines its chemical and … Its electron configuration is … 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl... That means there are 17 electrons in a.

Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds.. .. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific.

That means there are 17 electrons in a. The nucleus consists of 17 protons (red) and 18 neutrons (blue). The stability of an element's outer electrons determines its chemical and … 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. Chlorine is a diatomic molecule and contains only two chlorine atoms. It is a halogen (17 th group) in the 3 rd period of the periodic table. How many electrons are in each energy level of the bohr model for chlorine? Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. The atomic number of chlorine is 17; 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl... 22.05.2021 · what is the atomic structure for chlorine?
21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. .. Its electron configuration is …

How many electrons are in each energy level of the bohr model for chlorine? It is a halogen (17 th group) in the 3 rd period of the periodic table. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. The nucleus consists of 17 protons (red) and 18 neutrons (blue). 22.05.2021 · what is the atomic structure for chlorine? Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds.

The atomic number of chlorine is 17; Thus, it has seventeen protons and seventeen electrons. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. The nucleus consists of 17 protons (red) and 18 neutrons (blue). 17), the most common isotope of this element. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. How to draw the atomic model for chlorine. Its electron configuration is … 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl.. Its electron configuration is …

It is very easy to draw cl2 lewis structure... It is very easy to draw cl2 lewis structure.. The stability of an element's outer electrons determines its chemical and …

Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. How many electrons are in each energy level of the bohr model for chlorine? 22.05.2021 · what is the atomic structure for chlorine? 17), the most common isotope of this element. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. How to draw the atomic model for chlorine. It is very easy to draw cl2 lewis structure. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific.. The stability of an element's outer electrons determines its chemical and …
The nucleus consists of 17 protons (red) and 18 neutrons (blue).. The nucleus consists of 17 protons (red) and 18 neutrons (blue). How many electrons are in each energy level of the bohr model for chlorine? This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. The atomic number of chlorine is 17;. It is very easy to draw cl2 lewis structure.

05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. . The nucleus consists of 17 protons (red) and 18 neutrons (blue).

How to draw the atomic model for chlorine.. How to draw the atomic model for chlorine. 17), the most common isotope of this element. 22.05.2021 · what is the atomic structure for chlorine? Chlorine is a diatomic molecule and contains only two chlorine atoms.. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).
21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization... It is a halogen (17 th group) in the 3 rd period of the periodic table. It is very easy to draw cl2 lewis structure. The stability of an element's outer electrons determines its chemical and … The nucleus consists of 17 protons (red) and 18 neutrons (blue). How many electrons are in each energy level of the bohr model for chlorine? That means there are 17 electrons in a. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. Chlorine is a diatomic molecule and contains only two chlorine atoms. The atomic number of chlorine is 17;

Chlorine is a diatomic molecule and contains only two chlorine atoms. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization.

17), the most common isotope of this element. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. Chlorine is a diatomic molecule and contains only two chlorine atoms. Thus, it has seventeen protons and seventeen electrons. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. The nucleus consists of 17 protons (red) and 18 neutrons (blue).. It is a halogen (17 th group) in the 3 rd period of the periodic table.

Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure... Chlorine is a diatomic molecule and contains only two chlorine atoms. How to draw the atomic model for chlorine. Thus, it has seventeen protons and seventeen electrons.

It is very easy to draw cl2 lewis structure... It is very easy to draw cl2 lewis structure. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. How to draw the atomic model for chlorine. Its electron configuration is … Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure.

The atomic number of chlorine is 17; Its electron configuration is … 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization.. Chlorine is a diatomic molecule and contains only two chlorine atoms.

Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. The atomic number of chlorine is 17;. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds.

05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. That means there are 17 electrons in a. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. 22.05.2021 · what is the atomic structure for chlorine? 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. How to draw the atomic model for chlorine. The nucleus consists of 17 protons (red) and 18 neutrons (blue).

Its electron configuration is ….. How many electrons are in each energy level of the bohr model for chlorine? 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. It is a halogen (17 th group) in the 3 rd period of the periodic table. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. 22.05.2021 · what is the atomic structure for chlorine?. Thus, it has seventeen protons and seventeen electrons.

Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds.. How many electrons are in each energy level of the bohr model for chlorine? Thus, it has seventeen protons and seventeen electrons. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. The atomic number of chlorine is 17; Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. Its electron configuration is …. That means there are 17 electrons in a.

That means there are 17 electrons in a... . 22.05.2021 · what is the atomic structure for chlorine?

That means there are 17 electrons in a.. 17), the most common isotope of this element. How many electrons are in each energy level of the bohr model for chlorine? The nucleus consists of 17 protons (red) and 18 neutrons (blue). 22.05.2021 · what is the atomic structure for chlorine? It is a halogen (17 th group) in the 3 rd period of the periodic table. Its electron configuration is …. Its electron configuration is …

21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. . It is a halogen (17 th group) in the 3 rd period of the periodic table.

Chlorine is a diatomic molecule and contains only two chlorine atoms. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The atomic number of chlorine is 17; Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. The nucleus consists of 17 protons (red) and 18 neutrons (blue). 22.05.2021 · what is the atomic structure for chlorine? Chlorine is a diatomic molecule and contains only two chlorine atoms. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure.

Chlorine is a diatomic molecule and contains only two chlorine atoms. That means there are 17 electrons in a. The atomic number of chlorine is 17; Chlorine is a diatomic molecule and contains only two chlorine atoms.

17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Chlorine is a diatomic molecule and contains only two chlorine atoms.

The nucleus consists of 17 protons (red) and 18 neutrons (blue)... It is very easy to draw cl2 lewis structure. Chlorine is a diatomic molecule and contains only two chlorine atoms... 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific.

Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds... The atomic number of chlorine is 17; Its electron configuration is … Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. The stability of an element's outer electrons determines its chemical and … Thus, it has seventeen protons and seventeen electrons. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. 17), the most common isotope of this element.. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

Thus, it has seventeen protons and seventeen electrons.. It is very easy to draw cl2 lewis structure. It is a halogen (17 th group) in the 3 rd period of the periodic table. How to draw the atomic model for chlorine. 22.05.2021 · what is the atomic structure for chlorine? How many electrons are in each energy level of the bohr model for chlorine? Chlorine is a diatomic molecule and contains only two chlorine atoms. 17), the most common isotope of this element.

22.05.2021 · what is the atomic structure for chlorine? . The nucleus consists of 17 protons (red) and 18 neutrons (blue).

Thus, it has seventeen protons and seventeen electrons.. How to draw the atomic model for chlorine. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. The atomic number of chlorine is 17;

That means there are 17 electrons in a.. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds.

Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure.. How many electrons are in each energy level of the bohr model for chlorine?

Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 17), the most common isotope of this element.

Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure... The atomic number of chlorine is 17; How to draw the atomic model for chlorine. How to draw the atomic model for chlorine.

It is very easy to draw cl2 lewis structure. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. The atomic number of chlorine is 17; 22.05.2021 · what is the atomic structure for chlorine? Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. It is a halogen (17 th group) in the 3 rd period of the periodic table. The nucleus consists of 17 protons (red) and 18 neutrons (blue). Chlorine is a diatomic molecule and contains only two chlorine atoms. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. It is very easy to draw cl2 lewis structure.

Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. The atomic number of chlorine is 17; 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. The stability of an element's outer electrons determines its chemical and … How many electrons are in each energy level of the bohr model for chlorine? It is a halogen (17 th group) in the 3 rd period of the periodic table. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. 22.05.2021 · what is the atomic structure for chlorine? 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. That means there are 17 electrons in a. How many electrons are in each energy level of the bohr model for chlorine?

It is very easy to draw cl2 lewis structure. How many electrons are in each energy level of the bohr model for chlorine? The stability of an element's outer electrons determines its chemical and …

05.11.2011 · chlorine is an element in the periodic table which is denoted by cl.. 22.05.2021 · what is the atomic structure for chlorine? That means there are 17 electrons in a. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl.

17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. The stability of an element's outer electrons determines its chemical and …. 17), the most common isotope of this element.

It is very easy to draw cl2 lewis structure... 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The stability of an element's outer electrons determines its chemical and … It is very easy to draw cl2 lewis structure. How many electrons are in each energy level of the bohr model for chlorine?

22.05.2015 · the atomic model for chlorine is commonly represented in a very specific... How many electrons are in each energy level of the bohr model for chlorine? 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. It is very easy to draw cl2 lewis structure. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. Chlorine is a diatomic molecule and contains only two chlorine atoms. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. The nucleus consists of 17 protons (red) and 18 neutrons (blue). How to draw the atomic model for chlorine. That means there are 17 electrons in a. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure.

05.11.2011 · chlorine is an element in the periodic table which is denoted by cl... 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). It is a halogen (17 th group) in the 3 rd period of the periodic table. The nucleus consists of 17 protons (red) and 18 neutrons (blue). 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. 17), the most common isotope of this element. 22.05.2021 · what is the atomic structure for chlorine?.. Chlorine is a diatomic molecule and contains only two chlorine atoms.

22.05.2021 · what is the atomic structure for chlorine? 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. That means there are 17 electrons in a.

Chlorine is a diatomic molecule and contains only two chlorine atoms.. It is very easy to draw cl2 lewis structure. The stability of an element's outer electrons determines its chemical and … Chlorine is a diatomic molecule and contains only two chlorine atoms. 17), the most common isotope of this element. How to draw the atomic model for chlorine. That means there are 17 electrons in a. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. How to draw the atomic model for chlorine.

That means there are 17 electrons in a. The nucleus consists of 17 protons (red) and 18 neutrons (blue). Its electron configuration is … Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. Chlorine is a diatomic molecule and contains only two chlorine atoms. How many electrons are in each energy level of the bohr model for chlorine?

22.05.2021 · what is the atomic structure for chlorine?. It is very easy to draw cl2 lewis structure. It is a halogen (17 th group) in the 3 rd period of the periodic table.. 22.05.2021 · what is the atomic structure for chlorine?

21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. That means there are 17 electrons in a. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Its electron configuration is … It is a halogen (17 th group) in the 3 rd period of the periodic table. 17), the most common isotope of this element. It is very easy to draw cl2 lewis structure. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. How many electrons are in each energy level of the bohr model for chlorine?. 17), the most common isotope of this element.

Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure.. How many electrons are in each energy level of the bohr model for chlorine? The atomic number of chlorine is 17; That means there are 17 electrons in a. How to draw the atomic model for chlorine. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. It is a halogen (17 th group) in the 3 rd period of the periodic table. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. Its electron configuration is … 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. It is a halogen (17 th group) in the 3 rd period of the periodic table.

How to draw the atomic model for chlorine... 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. The stability of an element's outer electrons determines its chemical and … Its electron configuration is … Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. How many electrons are in each energy level of the bohr model for chlorine? 17), the most common isotope of this element. 22.05.2021 · what is the atomic structure for chlorine?. How to draw the atomic model for chlorine.

22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. Its electron configuration is … 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. It is a halogen (17 th group) in the 3 rd period of the periodic table. Thus, it has seventeen protons and seventeen electrons. It is very easy to draw cl2 lewis structure. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. That means there are 17 electrons in a. How many electrons are in each energy level of the bohr model for chlorine?. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds.

The nucleus consists of 17 protons (red) and 18 neutrons (blue).. This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization.. Its electron configuration is …

How many electrons are in each energy level of the bohr model for chlorine? How many electrons are in each energy level of the bohr model for chlorine? That means there are 17 electrons in a. How to draw the atomic model for chlorine. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. The atomic number of chlorine is 17; Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. 22.05.2021 · what is the atomic structure for chlorine? 22.05.2021 · what is the atomic structure for chlorine?
21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. How to draw the atomic model for chlorine. The atomic number of chlorine is 17; Its electron configuration is … 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. 05.11.2011 · chlorine is an element in the periodic table which is denoted by cl.. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization.

05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. It is a halogen (17 th group) in the 3 rd period of the periodic table. Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. Its electron configuration is ….. 22.05.2021 · what is the atomic structure for chlorine?

How many electrons are in each energy level of the bohr model for chlorine?. It is a halogen (17 th group) in the 3 rd period of the periodic table. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 17 protons (red) and 18 neutrons (blue). Its electron configuration is … It is very easy to draw cl2 lewis structure.. 22.05.2015 · the atomic model for chlorine is commonly represented in a very specific.

How many electrons are in each energy level of the bohr model for chlorine? It is very easy to draw cl2 lewis structure. Thus, it has seventeen protons and seventeen electrons. 17 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Chlorine is a diatomic molecule and contains only two chlorine atoms. How many electrons are in each energy level of the bohr model for chlorine?

It is a halogen (17 th group) in the 3 rd period of the periodic table. How many electrons are in each energy level of the bohr model for chlorine? 17), the most common isotope of this element.. It is a halogen (17 th group) in the 3 rd period of the periodic table.

22.05.2015 · the atomic model for chlorine is commonly represented in a very specific. 21.11.2021 · moreover, from the lewis structure, it is clear that the chlorine atom has tetrahedral electron geometry by which it has sp3 hybridization.

It is a halogen (17 th group) in the 3 rd period of the periodic table. . Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure.

It is a halogen (17 th group) in the 3 rd period of the periodic table. Thus, it has seventeen protons and seventeen electrons.

The stability of an element's outer electrons determines its chemical and …. How many electrons are in each energy level of the bohr model for chlorine? Thus, it has seventeen protons and seventeen electrons. 17), the most common isotope of this element. The atomic number of chlorine is 17; This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. That means there are 17 electrons in a.. It is very easy to draw cl2 lewis structure.

That means there are 17 electrons in a... This reactivity in biological systems makes it difficult to study the pharmacokinetics of chlorine and to separate the effects of chlorine from those of the chlorine compounds and metabolites. How many electrons are in each energy level of the bohr model for chlorine? Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure. Thus, it has seventeen protons and seventeen electrons. It is very easy to draw cl2 lewis structure. It is a halogen (17 th group) in the 3 rd period of the periodic table.
Chlorine, as chlorine gas, chlorite ion, and hypochlorite, is a strong oxidant that readily reacts with organic molecules to produce a variety of chlorinated compounds. Moreover, the presence of an equal number of valence electrons on both chlorine and oxygen atoms further gives hypochlorite a linear structure.

05.11.2011 · chlorine is an element in the periodic table which is denoted by cl. The atomic number of chlorine is 17; Chlorine is a diatomic molecule and contains only two chlorine atoms. How many electrons are in each energy level of the bohr model for chlorine? That means there are 17 electrons in a. 22.05.2021 · what is the atomic structure for chlorine? Thus, it has seventeen protons and seventeen electrons. Its electron configuration is … The nucleus consists of 17 protons (red) and 18 neutrons (blue). It is very easy to draw cl2 lewis structure.. It is very easy to draw cl2 lewis structure.
