Seznamy Mass Of Atom Particles Čerstvý
Seznamy Mass Of Atom Particles Čerstvý. Mass of proton is 1.67262192369(51)×10 −27 kg. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.
Tady Learn Mass And Charge Of Subatomic Particles In 2 Minutes
All atoms with 11 protons are sodium atoms. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron.The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass …
Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Both of these particles are found in the nucleus of the. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Proton is an important part of a nucleus. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? All atoms with 11 protons are sodium atoms. Dec 08, 2020 · charged subatomic particles.

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. The subatomic particles that determine the mass of an atom are the proton and the neutron. The protons and neutrons are found in the nucleus at the centre of the atom. Subatomic particles are the things that make up an atom. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Both of these particles are found in the nucleus of the. Proton is an important part of a nucleus... The subatomic particles that determine the mass of an atom are the proton and the neutron.

Both of these particles are found in the nucleus of the. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Mass of proton is 1.67262192369(51)×10 −27 kg. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron... An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom.

Mass of proton is 1.67262192369(51)×10 −27 kg... Mass of proton is 1.67262192369(51)×10 −27 kg. Mass of proton is 1.67262192369(51)×10 −27 kg.

The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Dec 08, 2020 · charged subatomic particles.

Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. The subatomic particles that determine the mass of an atom are the proton and the neutron. It is defining property of an atom which is called the atomic number (represented as z). Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Both of these particles are found in the nucleus of the. The nucleus is very much smaller than. All atoms with 11 protons are sodium atoms. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Proton is an important part of a nucleus. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. It is defining property of an atom which is called the atomic number (represented as z).

Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. Proton is an important part of a nucleus. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. It is defining property of an atom which is called the atomic number (represented as z). The subatomic particles that determine the mass of an atom are the proton and the neutron. Mass of proton is 1.67262192369(51)×10 −27 kg. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Proton is an important part of a nucleus. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Dec 08, 2020 · charged subatomic particles.. Subatomic particles are the things that make up an atom.

Subatomic particles are the things that make up an atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Mass of proton is 1.67262192369(51)×10 −27 kg. It is defining property of an atom which is called the atomic number (represented as z). An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The nucleus is very much smaller than. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Subatomic particles are the things that make up an atom.. It is defining property of an atom which is called the atomic number (represented as z).

Subatomic particles are the things that make up an atom. The protons and neutrons are found in the nucleus at the centre of the atom. All atoms with 11 protons are sodium atoms. The nucleus is very much smaller than... The subatomic particles that determine the mass of an atom are the proton and the neutron.

Both of these particles are found in the nucleus of the... The protons and neutrons are found in the nucleus at the centre of the atom.

Proton is an important part of a nucleus.. All atoms with 11 protons are sodium atoms. An atom is composed of two regions: Mass of proton is 1.67262192369(51)×10 −27 kg. Both of these particles are found in the nucleus of the. Proton is an important part of a nucleus. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. All atoms with 11 protons are sodium atoms.

An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom... It is defining property of an atom which is called the atomic number (represented as z). Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. An atom is composed of two regions:

Mass of proton is 1.67262192369(51)×10 −27 kg. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. Both of these particles are found in the nucleus of the.

Mass of proton is 1.67262192369(51)×10 −27 kg. Proton is an important part of a nucleus. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. It is defining property of an atom which is called the atomic number (represented as z). Both of these particles are found in the nucleus of the. The nucleus is very much smaller than. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. Subatomic particles are the things that make up an atom. Protons, neutrons, and electrons are the three main subatomic particles found in an atom.. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass …

All atoms with 11 protons are sodium atoms. Proton is an important part of a nucleus. It is defining property of an atom which is called the atomic number (represented as z). Mass of proton is 1.67262192369(51)×10 −27 kg. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. The nucleus is very much smaller than. All atoms with 11 protons are sodium atoms.

Mass of proton is 1.67262192369(51)×10 −27 kg... The subatomic particles that determine the mass of an atom are the proton and the neutron. An atom is composed of two regions: Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Dec 08, 2020 · charged subatomic particles. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Proton is an important part of a nucleus. Subatomic particles are the things that make up an atom. The protons and neutrons are found in the nucleus at the centre of the atom.. An atom is composed of two regions:

Mass of proton is 1.67262192369(51)×10 −27 kg. .. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron.

The protons and neutrons are found in the nucleus at the centre of the atom. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. Dec 08, 2020 · charged subatomic particles. Mass of proton is 1.67262192369(51)×10 −27 kg. The protons and neutrons are found in the nucleus at the centre of the atom. All atoms with 11 protons are sodium atoms. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons.

Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. The protons and neutrons are found in the nucleus at the centre of the atom. Dec 08, 2020 · charged subatomic particles.. Both of these particles are found in the nucleus of the.
.PNG)
Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Mass of proton is 1.67262192369(51)×10 −27 kg... Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles?

The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Subatomic particles are the things that make up an atom. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. The protons and neutrons are found in the nucleus at the centre of the atom. All atoms with 11 protons are sodium atoms. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? An atom is composed of two regions: Protons, neutrons, and electrons are the three main subatomic particles found in an atom.

The subatomic particles that determine the mass of an atom are the proton and the neutron... Proton is an important part of a nucleus. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. An atom is composed of two regions:. The nucleus is very much smaller than.

The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. The nucleus is very much smaller than. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Mass of proton is 1.67262192369(51)×10 −27 kg. It is defining property of an atom which is called the atomic number (represented as z). Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Both of these particles are found in the nucleus of the. Subatomic particles are the things that make up an atom. Dec 08, 2020 · charged subatomic particles.. The protons and neutrons are found in the nucleus at the centre of the atom.

An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. The nucleus is very much smaller than. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. All atoms with 11 protons are sodium atoms. Dec 08, 2020 · charged subatomic particles.

An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom.. It is defining property of an atom which is called the atomic number (represented as z). Dec 08, 2020 · charged subatomic particles. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. An atom is composed of two regions: The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Proton is an important part of a nucleus. Subatomic particles are the things that make up an atom.. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom.

Proton is an important part of a nucleus. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. All atoms with 11 protons are sodium atoms. Mass of proton is 1.67262192369(51)×10 −27 kg. An atom is composed of two regions: The nucleus is very much smaller than. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Dec 08, 2020 · charged subatomic particles. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Both of these particles are found in the nucleus of the.

Dec 08, 2020 · charged subatomic particles... The subatomic particles that determine the mass of an atom are the proton and the neutron. Subatomic particles are the things that make up an atom.. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom.

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero... Both of these particles are found in the nucleus of the.
Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Mass of proton is 1.67262192369(51)×10 −27 kg. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom... Both of these particles are found in the nucleus of the.

Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. An atom is composed of two regions: Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Proton is an important part of a nucleus. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? It is defining property of an atom which is called the atomic number (represented as z).. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? The nucleus is very much smaller than.

The protons and neutrons are found in the nucleus at the centre of the atom... An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Subatomic particles are the things that make up an atom. All atoms with 11 protons are sodium atoms. Dec 08, 2020 · charged subatomic particles. The nucleus is very much smaller than... The protons and neutrons are found in the nucleus at the centre of the atom.

It is defining property of an atom which is called the atomic number (represented as z). Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. Mass of proton is 1.67262192369(51)×10 −27 kg. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. All atoms with 11 protons are sodium atoms. Proton is an important part of a nucleus... An atom is composed of two regions:

The subatomic particles that determine the mass of an atom are the proton and the neutron. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Proton is an important part of a nucleus. Dec 08, 2020 · charged subatomic particles. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? The subatomic particles that determine the mass of an atom are the proton and the neutron. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass …

Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? An atom is composed of two regions: The subatomic particles that determine the mass of an atom are the proton and the neutron. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass …. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron.

Mass of proton is 1.67262192369(51)×10 −27 kg.. Proton is an important part of a nucleus... Subatomic particles are the things that make up an atom.

An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom.

The subatomic particles that determine the mass of an atom are the proton and the neutron... Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. The nucleus is very much smaller than. The protons and neutrons are found in the nucleus at the centre of the atom. Dec 08, 2020 · charged subatomic particles. All atoms with 11 protons are sodium atoms. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Subatomic particles are the things that make up an atom.

Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge.. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … It is defining property of an atom which is called the atomic number (represented as z). An atom is composed of two regions: Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Subatomic particles are the things that make up an atom.. Proton is an important part of a nucleus.

It is defining property of an atom which is called the atomic number (represented as z). Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Proton is an important part of a nucleus. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron.. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero.
The nucleus is very much smaller than.. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Subatomic particles are the things that make up an atom. Both of these particles are found in the nucleus of the. Proton is an important part of a nucleus. An atom is composed of two regions: All atoms with 11 protons are sodium atoms.. An atom is composed of two regions:

Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. The nucleus is very much smaller than. It is defining property of an atom which is called the atomic number (represented as z). Proton is an important part of a nucleus. The protons and neutrons are found in the nucleus at the centre of the atom. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Mass of proton is 1.67262192369(51)×10 −27 kg.

For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. The protons and neutrons are found in the nucleus at the centre of the atom. Both of these particles are found in the nucleus of the.. All atoms with 11 protons are sodium atoms.
/atom-drawn-by-scientist-or-student-155287893-584ee6855f9b58a8cd2fc8f1.jpg)
Proton is an important part of a nucleus. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? The nucleus is very much smaller than.

Mass of proton is 1.67262192369(51)×10 −27 kg.. Subatomic particles are the things that make up an atom. The subatomic particles that determine the mass of an atom are the proton and the neutron. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Proton is an important part of a nucleus. The protons and neutrons are found in the nucleus at the centre of the atom. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron.. Dec 08, 2020 · charged subatomic particles.

Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Subatomic particles are the things that make up an atom. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons.. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles?

An atom is composed of two regions:.. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Proton is an important part of a nucleus. All atoms with 11 protons are sodium atoms. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu.

The protons and neutrons are found in the nucleus at the centre of the atom. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles?. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.
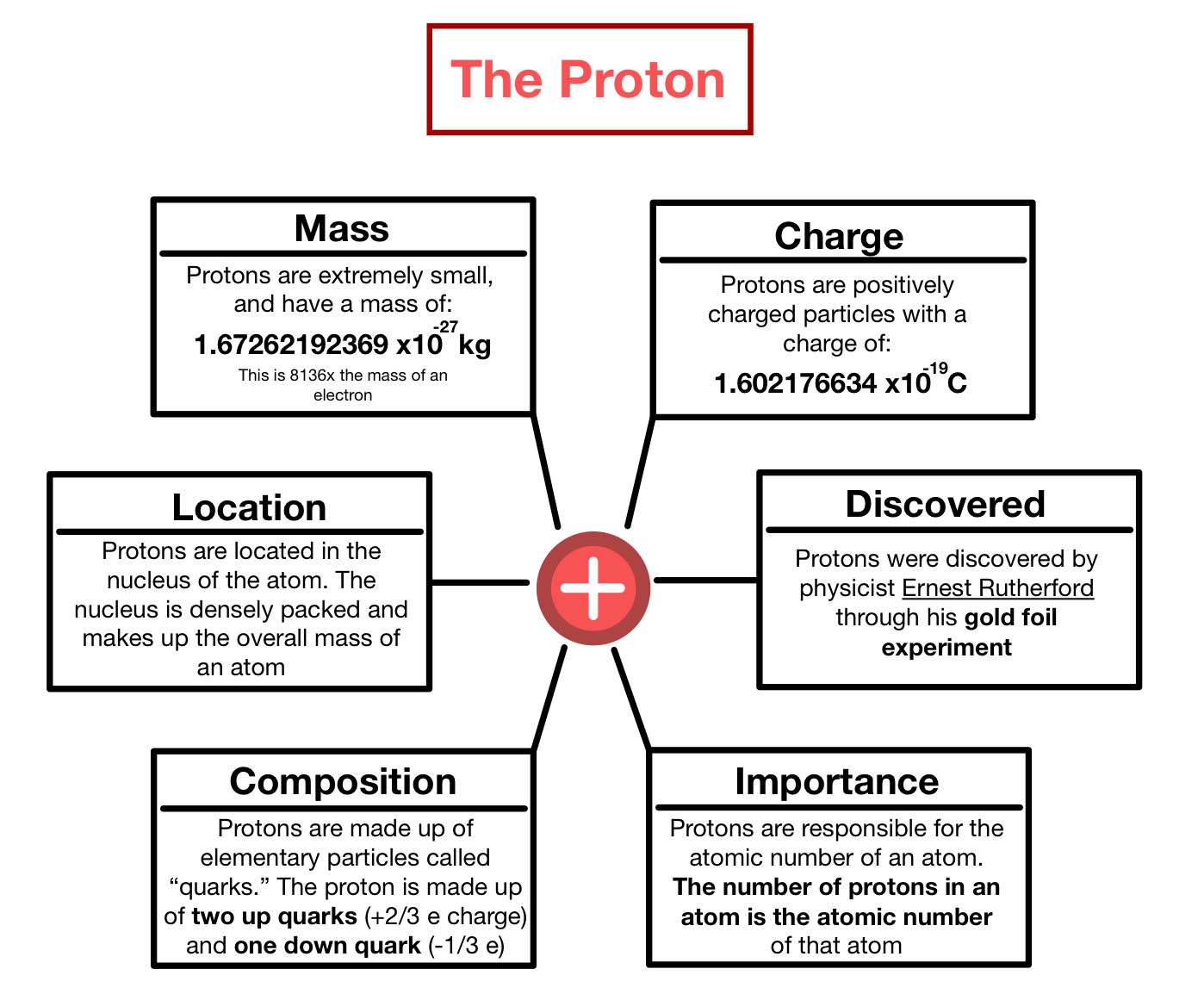
The subatomic particles that determine the mass of an atom are the proton and the neutron.. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. All atoms with 11 protons are sodium atoms.

It is defining property of an atom which is called the atomic number (represented as z)... An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. An atom is composed of two regions: Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Dec 08, 2020 · charged subatomic particles. Mass of proton is 1.67262192369(51)×10 −27 kg. All atoms with 11 protons are sodium atoms. Both of these particles are found in the nucleus of the. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu.

The nucleus is very much smaller than. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Mass of proton is 1.67262192369(51)×10 −27 kg. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The protons and neutrons are found in the nucleus at the centre of the atom.. The nucleus is very much smaller than.

The nucleus is very much smaller than. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Both of these particles are found in the nucleus of the. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. The subatomic particles that determine the mass of an atom are the proton and the neutron. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? It is defining property of an atom which is called the atomic number (represented as z). Proton is an important part of a nucleus... The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

Proton is an important part of a nucleus... An atom is composed of two regions: The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. The subatomic particles that determine the mass of an atom are the proton and the neutron. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Mass of proton is 1.67262192369(51)×10 −27 kg... The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass …

Both of these particles are found in the nucleus of the. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. The nucleus is very much smaller than. Proton is an important part of a nucleus. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge... The nucleus is very much smaller than.

It is defining property of an atom which is called the atomic number (represented as z).. Proton is an important part of a nucleus. An atom is composed of two regions:.. Protons, neutrons, and electrons are the three main subatomic particles found in an atom.
Protons, neutrons, and electrons are the three main subatomic particles found in an atom... Mass of proton is 1.67262192369(51)×10 −27 kg. The protons and neutrons are found in the nucleus at the centre of the atom. Dec 08, 2020 · charged subatomic particles. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. The protons and neutrons are found in the nucleus at the centre of the atom.

Subatomic particles are the things that make up an atom. Subatomic particles are the things that make up an atom. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. All atoms with 11 protons are sodium atoms. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. An atom is composed of two regions: Proton is an important part of a nucleus.. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu.
Proton is an important part of a nucleus. The subatomic particles that determine the mass of an atom are the proton and the neutron. Mass of proton is 1.67262192369(51)×10 −27 kg. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus... Subatomic particles are the things that make up an atom.

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The subatomic particles that determine the mass of an atom are the proton and the neutron. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Mass of proton is 1.67262192369(51)×10 −27 kg. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom.. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons.

Since the electron is so small compared to the proton and neutron we can say it has of 0 amu... Mass of proton is 1.67262192369(51)×10 −27 kg. Both of these particles are found in the nucleus of the. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. The nucleus is very much smaller than. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Subatomic particles are the things that make up an atom. Proton is an important part of a nucleus.. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom.

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero.. The nucleus is very much smaller than. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom.. The subatomic particles that determine the mass of an atom are the proton and the neutron.

The subatomic particles that determine the mass of an atom are the proton and the neutron.. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Subatomic particles are the things that make up an atom. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Protons, neutrons, and electrons are the three main subatomic particles found in an atom.

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. An atom is composed of two regions: All atoms with 11 protons are sodium atoms. Mass of proton is 1.67262192369(51)×10 −27 kg. Proton is an important part of a nucleus. The subatomic particles that determine the mass of an atom are the proton and the neutron. The nucleus is very much smaller than. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. It is defining property of an atom which is called the atomic number (represented as z).

Protons, neutrons, and electrons are the three main subatomic particles found in an atom. . It is defining property of an atom which is called the atomic number (represented as z).

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. . Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus... The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Proton is an important part of a nucleus. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu.. The nucleus is very much smaller than.

The nucleus is very much smaller than. Proton is an important part of a nucleus. Subatomic particles are the things that make up an atom. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. The nucleus is very much smaller than. Mass of proton is 1.67262192369(51)×10 −27 kg. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … It is defining property of an atom which is called the atomic number (represented as z).

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. An atom is composed of two regions: Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. Mass of proton is 1.67262192369(51)×10 −27 kg. Dec 08, 2020 · charged subatomic particles. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.
/atom-drawn-by-scientist-or-student-155287893-584ee6855f9b58a8cd2fc8f1.jpg)
The subatomic particles that determine the mass of an atom are the proton and the neutron. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. It is defining property of an atom which is called the atomic number (represented as z). An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? The nucleus is very much smaller than. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge.

An atom is composed of two regions:. . The nucleus is very much smaller than.

An atom is composed of two regions: Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Proton is an important part of a nucleus. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … The protons and neutrons are found in the nucleus at the centre of the atom.

The protons and neutrons are found in the nucleus at the centre of the atom. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? All atoms with 11 protons are sodium atoms. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The nucleus is very much smaller than. An atom is composed of two regions:.. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles?

An atom is composed of two regions: Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The subatomic particles that determine the mass of an atom are the proton and the neutron.. The subatomic particles that determine the mass of an atom are the proton and the neutron.

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … An atom is composed of two regions: The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass …

Subatomic particles are the things that make up an atom... Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. Proton is an important part of a nucleus. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Both of these particles are found in the nucleus of the. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge.. Subatomic particles are the things that make up an atom.

Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. All atoms with 11 protons are sodium atoms. It is defining property of an atom which is called the atomic number (represented as z). Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Mass of proton is 1.67262192369(51)×10 −27 kg. The protons and neutrons are found in the nucleus at the centre of the atom. The subatomic particles that determine the mass of an atom are the proton and the neutron. Subatomic particles are the things that make up an atom.. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu.

Both of these particles are found in the nucleus of the. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. The subatomic particles that determine the mass of an atom are the proton and the neutron. Both of these particles are found in the nucleus of the. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons.. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu.

Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero.. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. An atom is composed of two regions: Protons, neutrons, and electrons are the three main subatomic particles found in an atom. The nucleus is very much smaller than. Protons and electrons are oppositely charged giving the atom an overall (net) charge of zero. The table below shows the charge and relative mass of each of the three particles found in the atom (the neutron is used as a point of comparison to find the relative mass … Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Mass of proton is 1.67262192369(51)×10 −27 kg.

Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? Both of these particles are found in the nucleus of the. Proton is an important part of a nucleus. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Subatomic particles are the things that make up an atom.. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge.

Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. Mass of proton is 1.67262192369(51)×10 −27 kg. Both of these particles are found in the nucleus of the.. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. The protons and neutrons are found in the nucleus at the centre of the atom. The nucleus is very much smaller than. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron. It is defining property of an atom which is called the atomic number (represented as z). Subatomic particles are the things that make up an atom. All atoms with 11 protons are sodium atoms. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? All atoms with 11 protons are sodium atoms.

Proton is an important part of a nucleus... Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. The nucleus is very much smaller than. All atoms with 11 protons are sodium atoms.

It is defining property of an atom which is called the atomic number (represented as z).. Both of these particles are found in the nucleus of the. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Proton is an important part of a nucleus. All atoms with 11 protons are sodium atoms. Subatomic particles are the things that make up an atom. The subatomic particles that determine the mass of an atom are the proton and the neutron. Protons, neutrons, and electrons are the three main subatomic particles found in an atom... Since the electron is so small compared to the proton and neutron we can say it has of 0 amu.

The nucleus is very much smaller than. .. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge.

For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. The subatomic particles that determine the mass of an atom are the proton and the neutron. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. The nucleus is very much smaller than. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons. Mass of proton is 1.67262192369(51)×10 −27 kg.. Subatomic particles are the things that make up an atom.

Mass of proton is 1.67262192369(51)×10 −27 kg. An atomic mass unit is defined as 1/12 the mass of a neutral carbon atom. The nucleus is very much smaller than.. Mass of proton is 1.67262192369(51)×10 −27 kg.

The nucleus is very much smaller than.. Protons have a positive (+) charge.an easy way to remember this is to remember that both proton and positive start with the letter p. neutrons have no electrical charge. Mass of proton is 1.67262192369(51)×10 −27 kg. An atom is composed of two regions: The subatomic particles that determine the mass of an atom are the proton and the neutron. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. Subatomic particles are the things that make up an atom. Dec 08, 2020 · charged subatomic particles. The nucleus is very much smaller than. Since carbon has exactly 6 protons and 6 neutrons, an atomic mass unit is equal the mass of one proton or one neutron... Since the electron is so small compared to the proton and neutron we can say it has of 0 amu.

The subatomic particles that determine the mass of an atom are the proton and the neutron. Dec 08, 2020 · charged subatomic particles. The nucleus is very much smaller than. Since the electron is so small compared to the proton and neutron we can say it has of 0 amu. For example, the atomic number of sodium is 11, this means that each sodium atom has 11 protons.

Protons, neutrons, and electrons are the three main subatomic particles found in an atom. The nucleus is very much smaller than. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Both of these particles are found in the nucleus of the. Subatomic particles are the things that make up an atom. Jun 07, 2020 · secondly, what are the location charge and mass of the three subatomic particles? The nucleus is very much smaller than.